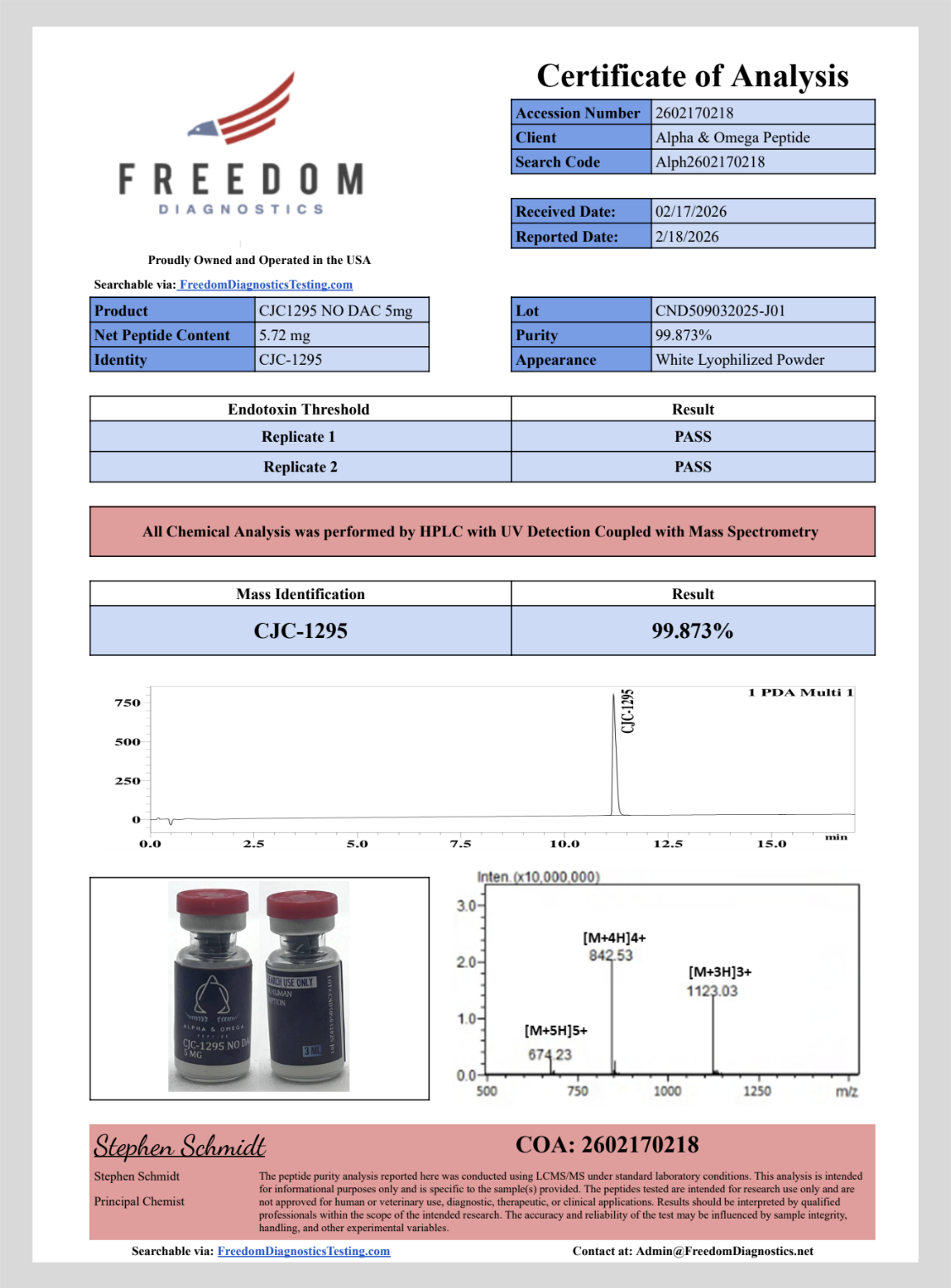
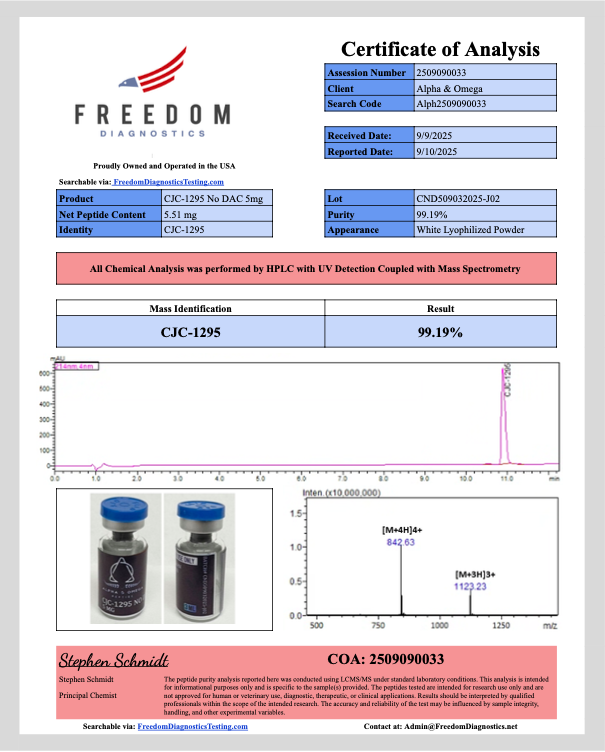
CJC-1295 (no DAC)
CJC-1295 (no DAC) is a research grade synthetic peptide developed for controlled laboratory investigation of growth hormone releasing hormone pathways. This non-DAC format allows research teams to evaluate timing dependent behavior within structured experimental windows. Because it excludes Drug Affinity Complex modification, it is often selected for study designs that prioritize tighter observation intervals, repeatable sampling, and protocol flexibility.
In RUO settings, compound selection depends on consistency, documentation, and reliable handling characteristics. Therefore, CJC-1295 (no DAC) is supplied in a lyophilized format to support stable storage prior to controlled reconstitution. When used within qualified laboratory environments, it fits well into workflows that require precise planning, clear recordkeeping, and repeatable procedures.
Non-DAC Structure for Timing Controlled Research
The “no DAC” designation indicates that this peptide does not include an extended binding modification. As a result, researchers can better align experimental timing with short window data collection, comparative models, and protocol variations. Additionally, this structure supports research designs that focus on pulse style evaluation, time course comparisons, and method development for analytical measurement.
Key Features for Laboratory Workflows
- Synthetic peptide supplied for research use only
- Non-DAC modified structure for timing specific study design
- Lyophilized powder format for controlled reconstitution procedures
- Suitable for structured in vitro evaluation and laboratory investigation
- Produced with quality focused processes appropriate for RUO handling
Research Applications and Protocol Fit
CJC-1295 (no DAC) is commonly integrated into research frameworks where timing, sampling, and comparison are central. For example, laboratories may use it while developing assay methods, evaluating pathway responses, or comparing non-DAC and DAC style formats under consistent conditions. Because timing variables can materially impact study outcomes, the non-DAC structure can help align observation windows with planned collection points and analytical readouts.
Moreover, the lyophilized presentation supports inventory control and lab routine standardization. This can simplify preparation steps when teams follow validated procedures for labeling, documentation, and controlled reconstitution. Consequently, researchers can maintain consistent handling practices across multiple runs and protocol iterations.
Handling and Storage Notes
Store CJC-1295 (no DAC) in a cool, dry environment prior to reconstitution. After reconstitution, follow standard laboratory storage protocols based on your internal procedures and study requirements. Always use proper sterile technique, calibrated equipment, and appropriate documentation for each preparation step. For best consistency, maintain a controlled workflow that includes lot tra